 CAPA (corrective and preventive actions) events with potential risk to patient safety.Ī good approach for risk management is using the BMX method, which includes hazard identification, risk estimation, risk control, risk evaluation and monitoring.each change (in a part) of a realization process of a released product, including changes to manufacturing sites and suppliers.discovery of mislabeled or non-conforming products.each change (in a part) of an already released device.each new medical device or derivative device.The risk management process is applicable for: 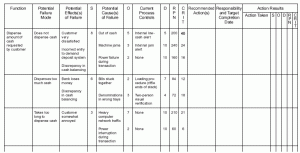
And, if a risk analysis for a product similar to yours is available, relevant and adequate, you can apply it to the study of your medical device. You must implement risk management in any product development project, from inception to completion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
